Opal - The Colorful Silica
Opal is most valued for the rich variety of colors it exhibits, their interplay in ordinary light, and their unusual range of fluorescence. These attributes have guaranteed its continuing value as a gemstone since pre-Roman times.
Lacking a true crystal structure, opal is a mineraloid. Nevertheless, nearly all historic reference sources classified it as a mineral, a category justified in part because some constituents of opal are now known to be microcrystalline forms of silica. Opal is an exception from IMA rules and it is classified as a true mineral.
The value placed on opal has fluctuated significantly, based on contemporary cultural beliefs. For example, during the Middle Ages, any opal that displayed the spectrum of colors associated with all other gemstones was considered an exceptionally lucky stone. Under bizarre circumstances, some believed it conferred invisibility on the wearer. On the other hand, the publication of Sir Walter Scott's Anne of Geierstein, in which an opal possessing supernatural powers is rendered colorless and impotent upon contact with holy water, caused a precipitous 50% drop in European sales of opal for the next 20 years. And in the early 20th century, Russians could believe that catching sight of an opal among items for sale was a reason to avoid making further purchases, for fear of provoking the evil eye.

Precious opal specimens can present one frustrating characteristic that will aggravate the jeweler attempting to fashion it, or the collector attempting to display it: Opal may NOT retain its integrity when dried out too quickly, or exposed to vibration, abrupt temperature changes or bright light!
Nomenclature
Our word opal has been linked to three possible sources, a Roman term (opalus), a Greek term (opallios), and a Sanskrit term (upala). The Roman usage appears to reflect a Latin term for precious stone. The Greek term is explained as an amalgamation of two meanings relating to seeing (a root for the English word opaque) and other (a root for the English word alias). The concept is that combining seeing with other is a good description for observing the color variations in opal.

However, regardless of the later Greek and Roman modifications of the term, it is clear that Roman references to the Sanskrit upala appear around 250 BC, and refer specifically to gemstones being supplied from traders in the Bosporus region of Asia, who identified their source in India.
CRYSTAL STRUCTURE OF OPAL
Opal is a form of amorphous (non-crystalline) silica with the formula SiO2.nH2O. Opal belongs to oxides or less often by some classification system into tectosilicates, but the silica portion of its composition is normally composed of one Si centered in a tetrahedron of 4 atoms of O. However, most forms of opal also contain a certain amount of water. The water content ranges from 3 % to 21 %.
Opal is conventionally classified into 2 main and 4 sub-categories:
Non-crystalline opal
- Opal-AG Amorphous-Gel (closely packed amorphous silica spheres)
- Opal-AN Amorphous Network (water-containing amorphous silica-glass)
Microcrystalline opal
- Opal-CT Cristobalite-Tridymite (spheres of opal are made up of tiny microcrystalline blades of cristobalite and tridymite)
- Opal-C Cristobalite (spheres of opal composed of cristobalite only)
Transitions between opal-AG, opal-CT and opal-C are common. In fact, recent research on opal confirmed some sort of ordered structure in many opals. Opal will be likely reclassified into several minerals in the future. Unfortunately, it is almost impossible to distinguish amorphous or cryptocrystalline forms of silica from each other - various opal and chalcedony forms are very similar and produce an astonishing variety of colors.

Diagenesis - the gradual sedimentary burial of rocks under heat and pressure - is believed to produce gradual transitions from the non-crystalline to microcrystalline forms of opal. The amorphous forms of silica tend to internally recrystallize into more stable forms. This process takes decades in human-made glass and millions of years in opals.
Opal is well-known for mimicking the forms of other minerals, as well as wood, shell or bone as pseudomorphs. Its usual habits run the gamut, including massive, botryoidal, nodular, earthy, reniform, vein-like and crusty. Opal does not produce any crystal forms.
PHYSICAL PROPERTIES OF OPAL
The color of opal covers the visible spectrum: Colorless, white, yellow, orange, red, purple, blue, green, gray, brown, and black. Banded and multicolored varieties are common. The mineral is vitreous, but may be pearly, resinous or waxy; variable in hardness (4.5 - 6.5), with a white streak, and a density between 1.98 and 2.25. Its fracture pattern is typically conchoidal, but may be splintery, uneven or irregular. It has no cleavage.

Play of color
Opal can display different colors when the light source is moved, when the stone is turned, or when viewed from different directions. These color flashes, which vary from stone to stone, are called schillers. Electron microscopy has clarified how this effect occurs. Tiny silica spheres are packed in an orderly arrangement. A light beam entering the array of spheres is diffracted and reflected out of the mineral in a variety of sheens and scintillations. Common (ordinary) opal does not create this effect, since the spheres are disordered and not systematically packed. Light entering such a specimen is not diffracted.

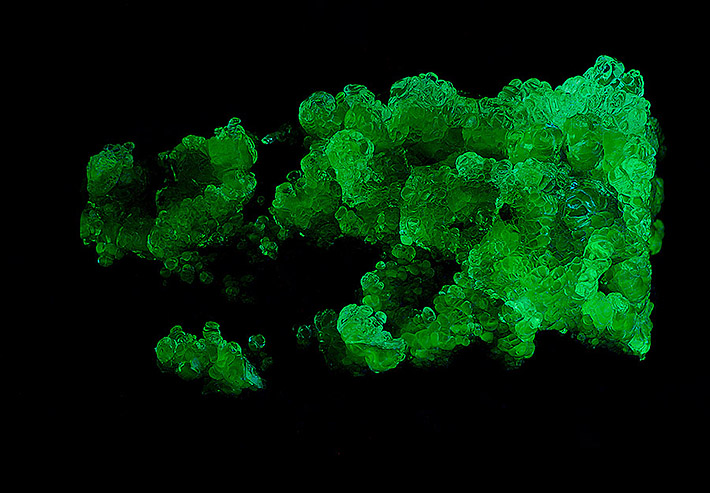
Opal Fluorescence
Opals are classic examples of minerals capable of fluorescing. Fluorescent minerals exhibit a color change when an energetic beam of ultraviolet light or X-rays of a specific wavelength causes emission of visible light. The human eye perceives this emitted light as fluorescence. The mineral will fluoresce as long as the incident light stimulus is applied.
Crazing of Opal
Opal is prone to a condition known as crazing. This is the phenomenon produced when a specimen develops internal and external cracks as a result of a variety of disturbances that, in other minerals, are negligible. Principal causes of crazing are:
- Allowing the sample to dry too quickly (removing it from its formerly damp environment)
- Exposing the sample to a sudden temperature change
- Exposing the sample to sudden bright light
- Vibrating the sample (during jewelry cutting and polishing)
Some opal specimens are known to be likely to craze, but the process is generally random, inconsistent and unpredictable. Experience has shown that some specimens can be stabilized by proceeding with the drying out process over a period of months or years.

One of the obvious reasons of crazing is a loss of water. So the overall chance of crazing depends mostly on water content and internal microstructure. Some localities produce high quality and very resistant opals while some others produce nice opals very prone to crazing. This is one of the reasons for the very high price of true Australian precious opals.
The crazing problem results in the inconvenient remedy of storing opal specimens underwater. This naturally reduces their susceptibility to crazing, but makes it equally hard to present them elegantly for viewing. To preserve the specimens intact, collectors need to avoid drying of opals or sudden change in temperature or light.
Similar minerals
Opal is generally not confused with any other mineral except chalcedony, a cryptocrystalline form of silica with a noticeably greater hardness (6.0 - 7.0).
FRAUDS AND SCAMS
Various precious opal frauds and scams are often present in the business, ranging from disreputable treatment and fake labels (opals from some localities are less prone to crazing) and extending to oiled, nail-polished or even completely faked opals.
Many dealers soak their opals to hide the crazing effects and lower the risk of crazing of low quality opals. Many even sell wet hydrophane as a precious opal. There is no problem if the origin and treatment of the opals is fully disclosed - which is often not the case, unfortunately.

Some dishonest dealers even create precious opals from plastic. These are of course much softer then true opal but can be hard to recognize for inexperienced buyers. Even more tricks are used in jewelry business.
ORIGIN OF OPAL
Opal forms from the alteration of high-silica volcanic tuffs, flows and siliceous marine sediments. Opals can precipitate from various hydrothermal fluids rich in silica. Opal-C, opal-CT and opal-AG are the most typical products in low-pressure and low-temperature environments, while opal-AN can originate from hot fluids.

Opals are present in huge quantities in certain serpentinites. These rocks are formed by hydration of mantle rocks called peridotites. The alteration process creates an excess of silica, which precipitates in the form of opal.
Common opals are abundant in various sedimentary and volcanic rocks. Opals in the metamorphic rocks are mostly limited to altered serpentinites or some weakly metamorphosed marine sediments. Opal does not occur as a magmatic mineral in igneous rocks but it might precipitate from hydrothermal fluids in the very late stage of magma crystallization.
APPLICATIONS
Industrial use of opal is mostly limited to diatomites - sedimentary silica rocks made of microscopic opal shells of Diatomeae marine plankton. Diatomites are used in lightweight construction materials, industrial filters, polishing materials, and special ceramics.

Opal holds very high value as a gemstone, the most prized of which are Black Opal, Precious Opal, and Fire Opal; all of these display play of colors. Mineral collectors and museums compete vigorously for the finest specimens. A subset of the collecting world involves just those specimens that fluoresce.
OPAL OCCURENCE
Opal is very common and occurs worldwide. Sedimentary opal like menilite or diatomite can even be an important rock forming mineral. The world-class sites for collecting opal are located in Australia, Mexico and the US.
Australia hosts many world-class opal localities: Andamooka, Coober Pedy and Mintabie (South Australia); Lightning Ridge and White Cliffs (New South Wales); Bull Creek, Hungerford, Opalton, Opalville, Quilpie and Yowah (Queensland).

Many precious opal localities are located in US: Last Chance, Kern County (California); Spencer area, Clark County (Idaho); Virgin Valley, Humboldt County (Nevada) and Opal Butte, Morrow County (Oregon).
Queretaro (Queretaro State) and Magdalena (Jalisco State) in Mexico are world-class sites for fire opal. Canadian precious opals come from Vernon, British Columbia. Other sites include Menze Gishe in Ethiopia, Piaui in Brazil Gracious and O Dios in Honduras.
World-class hyalite specimens are known from Valeč, Czech Republic. Strongly fluorescent and very aesthetic hyalites were recently discovered in Tarcal, Hungary.
OPAL VARIETES
Opal is plagued by a surfeit of prefixes and descriptive terms. A few of the terms, such Fire Opal and Hyalite, have received universal endorsement, but most are isolated names that have appeared only a few times in published reports or catalogs. The following is a web-based list of the many terms that have been applied:
Place names: Andamooka, Coober Pedy, Hungarian, Lightning Ridge, Nevada, Virgin Valley, White Cliffs, Yowah Nut.

Color names: Black, Cherry, Chloropal, Chrysopal, Claro, Common, Contra Luz, Crystal, Fire, Flash, Harlequin, Honey, Hyalite, Hydrophane, Jelly, Lemon, Mexican Fire, Milk, Pinfire, Precious, Precious Fire, Water, Wax, White.
Marking names: Banded, Boulder, Diatomite, Fiorite, Geyserite, Matrix, Moss, Onyx, Perlite, Pineapple, Seam, Tabasheer, Wood, Menilite.
Common opal
As the name suggests - common opal is the most abundant form of opal. Such opals are usually white, yellow, red, brown, grey or multicolored. Unlike precious opals, the common opals are not very prone to crazing and can survive for years without a change. Some sorts of common opals have their own variety names.

Precious opal
All forms of opals with play of colors are labeled as precious opal. There are many sub-varieties though: white opal has a white matrix, black opal is dark, fire opal is orange-red. Many precious opals are labeled after their locality: Lightning Ridge and White Cliff (NSW), Yowah Nut and Boulder (Queensland), Andamooka and Coober Pedy (South Australia), Nevada and Virgin Valley (Nevada), Claro (Mexico), and many others.
Precious opals (mostly of sedimentary origin) from Australia and Brazil are known to be very stable and resistant to crazing. On the other hand, precious opals (mostly of volcanic origin) from Ethiopia, Mexico and USA are infamous for their crazing and loss of play of colors.

The first precious opals were discovered in andesites in Dubník, Slovakia. However, gem-quality opals are mostly small and most of the opals at the deposit are low quality milky opal or hydrophane. Only few of the historic specimens retain their play of colors after 100 years or more.
Fire opal
Translucent yellow-orange or orange-red opals are often labeled as fire opal. The best quality fire opals show the play of colors and are known also as precious fire opal. The best fire opals definitely come from Mexico.

Hydrophane
The term hydrophane is used for opals of volcanic origin. The other use of this term refers to hydrophane's ability to absorb water. Many hydrophanes are white when dry and become translucent or even display play of colors when wet. Some may say that hydrophane is something between low quality precious opal and milky opal.

Hyalite
Hyalites are often formed within the cracks in rocks. Most common are those from lavas, but hyalites are known also as late stage hydrothermal minerals from pegmatites or filling fissures in marbles. Hyalite is usually colorless and translucent; less common are pale yellow or pale blue colors. Many hyalites show strong fluorescence in UV light.

Milk(y) opal
White opal with vitreous or resinous luster is sometimes called milk opal. Milk opal should not be confused with white opal (a form of precious opal). Unlike cachalong, milk opal is not that soft or chalky in appearance. Milk opals from serpentinites often contain dendrites.

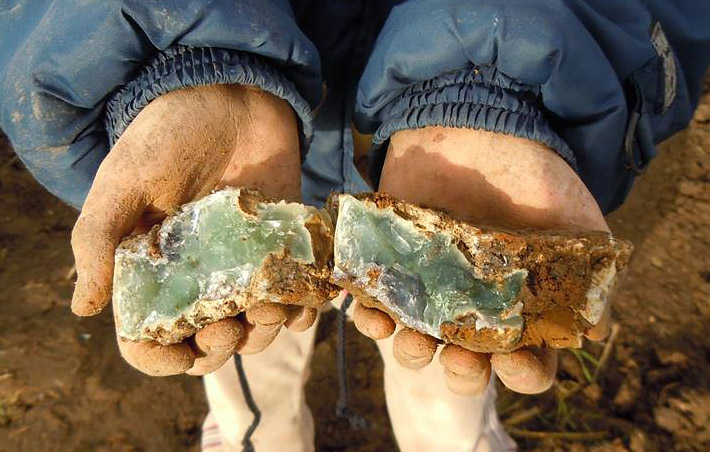
Prase opal
Many serpentinites contain some nickel or chromium in their minerals. When opals are formed during serpentinite alteration, nickel and chromium can color them apple green.

Moss/dendritic opal
Some opals contain black dendrites made of iron-manganese oxides. The dendrites can create highly aesthetic patterns in the white, yellow or green opals. Semi translucent opal with dendrites are very nice and highly popular for production of inexpensive cabochons.

Cachalong
Very low quality opaque form of chalky white colored opal is called cachalong. Such opal is usually soft and easily damaged; it can be mixed with magnesite in some serpentinites. Cachalong contains the lowest amount of water among all types of opal.
Wood opal
Some of the awesome fossil trees are made of opal. Such variety is often called a wood opal. It originates when fluids rich in silica circulate within porous rocks and precipitate opal within the buried tree trunks. Wood opal is common in many areas where tuffs and ash covered forests on the slopes of volcanos, and later volcanic fluids circulated within the rocks. Very old Paleozoic opalized fossil woods slowly recrystallized into chalcedony. Opal is not stable in geologic time and changes into chalcedony or quartz over hundreds of millions of years. Younger wood opal is usually associated with Tertiary volcanism.

Shell opal
As mentioned in the wood opal section, opal can replace fossils. Some sediments - especially in Australia - are famous for their opalized fossils. Some fossils are replaced with precious opal; some of the most famous are dinosaur bones or teeth fully replaced by high quality precious opal.

Diatomite
Despite their microscopic size, the Diatomeae create significant mass in the ocean volume. These tiny marine algae are responsible for the creation of 20-25 % of world oxygen production. They create silica shells, mostly composed of opal. When organisms die, the shells sink to the ocean floor, where they accumulate into thick layers and form silica rocks called diatomites.
Menilite
Menilite is a gray sedimentary from of opal, which forms nodules or layers in certain marine sedimentary rocks. Menilites are formed from diatomites during diagenesis - the gradual sedimentary burial of rocks under heat and pressure. The weight of the overburden produces pressure and highly porous and soft diatomites rearrange into more compact forms of opal or chalcedony.






Comments
Opal Auctions said:
25.05.2016 02:09:18This is a fantastic write up. Thank you for spreading the word about precious Opal. I am sharing this article on our Facebook page!