Feldspars - all about the plagioclases and K-feldspars
Feldspars are alumosilicates of alcalic metals and form a significant portion of many volcanic and metamorphic rocks. As such, they are one of the most important mineral groups on the Earths surface: some estimate that they form between 50-60 % of the Earths crust. Their content in most sedimentary rocks is much less significant.
Feldspars are mostly used in ceramics industry. Gem quality clear feldspars, moonstone and labradorite are used in jewelry. Labradorite rich rocks or amazonite granites are also used as decorative building stones.
Structure and Chemical Composition of Feldspars
Feldspars belong to the tectosilicates, sometimes referred also as network silicates. Their structure is based on three dimensional network of SiO4 tetrahedrons which are partially replaced by AlO4 tetrahedrons.
Feldspars form monoclinic or triclinic crystals. They are divided into groups of the disordered (high) and the ordered (low) feldspars. The high feldspars are mostly monoclinic and the low feldspars are usually triclinic. The order is related to randomness of the distribution of AlO4 tetrahedrons inside the basic SiO4 tetrahedrons network. The less regular distribution of Al in structure, the less ordered the feldspar. The order of the structure improves (becomes less random) with the decreasing temperature.

Size: 8 x 8 cm. Photo: Zbynek Burival
The chemical composition of feldspars includes alcalic metals, aluminum, silica and oxygen. The general chemical formula is a bit complicated (K,Na)1-x(Ca,Ba)x(Si3-xAl1+xO8). This complexity is caused by presence of both monovalent and divalent cations which require more complicated heterovalent substitution.
Generally, feldspars can be divided into three main groups:
- Plagioclase feldspars (Na or Ca): NaAlSi3O8 - CaAl2Si2O8 (albite and anorthite)
- Alcalic feldspars (K or Na): K/NaAlSi3O8 (albite and all K-feldspars)
- Barium feldspars (Ba): BaAl2Si2O8 (celsian)
Complete solid solution (miscibility) only occurs in the plagioclase group, between albite and anorthite. Albite and K-feldspars mix only at high temperatures. When the temperature drops, their structures do not mix anymore and form "worm-like" shapes in each other. Small pieces of albite in K-feldspar are called perthite, the reverse is antiperthite. By comparison, K-feldspars mix very well with Ba-feldspars.
The chemical resistance of feldspars is very limited. They are often replaced by younger minerals, usually micas or clays. Chemical weathering of K-feldspar-rich rocks often forms layers of kaolinite.
Physical Properties of Feldspars
The feldspars are usually pale colored - often white, light gray, pale yellow, pink or orange. Less common are dark grey, brown or even green colors. Very rare gemmy feldspars can be almost clear with usually yellow or clear color.

The Mohs hardness of most feldspars is 6; some can even reach 7. The specific gravity depends strongly on composition and varies between 2.5-2.9 g/cm3. Feldspars show perfect or very good cleavage in two dimensions and crystals can be easily damaged or split. The luster is vitreous, sub-vitreous or resinous.
Feldspars form shortly prismatic or tabular crystals, often twinned. The crystal twinning laws are called Carlsbad, Baveno or Manebach. Plagioclase often forms multiple twins (polysynthetic twinning).
Occurence
Feldspars are present in almost all magmatic and metamorphic rocks, but are less common in sedimentary rocks because of their low mechanical and chemical stability. Generally, magmatic rocks with more silica (granitoids, pegmatites, greisens) usually contain K-feldspars and albite. Rocks with less silica contain Ca-rich plagioclase and the most Si depleted rocks contain mostly minerals from foids group like nepheline or haüyne.
Pegmatites are definitely the most important feldspars source. Pegmatite is an extremely coarse grained rock of granitic composition which is usually pale in color. Pegmatites form veins and lenses and often have pockets full of excellent quality crystals. Because of their chemical evolution, they usually contain lot of both K-feldspars and albite, often in the form of very nice crystals, which can grow to sizes upwards of several meters in length.

Albite and K-feldspars occur also in a high temperature granite body cap called greisen. Greisens are complex rocks, integrating both magmatic and hydrothermal phenomena. Between the magmatic granite body and greisen cap is usually a layer of extremely coarse grained crystals of K-feldspar, quartz and zinnwaldite mica. This layer is called stockscheider and in some ways is quite similar to pegmatites. Some parts of a granite cap or the greisen itself can produce huge masses of albite. The event - when the albite replaces K-feldspars or other minerals - is called albitization and it is quite typical for greisens.
Other environments rich in very nice feldspar crystals are so called alpine clefts. Alpine clefts are fissures in rocks filled with low temperature hydrothermal minerals, typically quartz, various chlorites, hematite, rutile and anatase. Feldspars often found in Alpine clefts are pericline and adularia.
Plagioclase
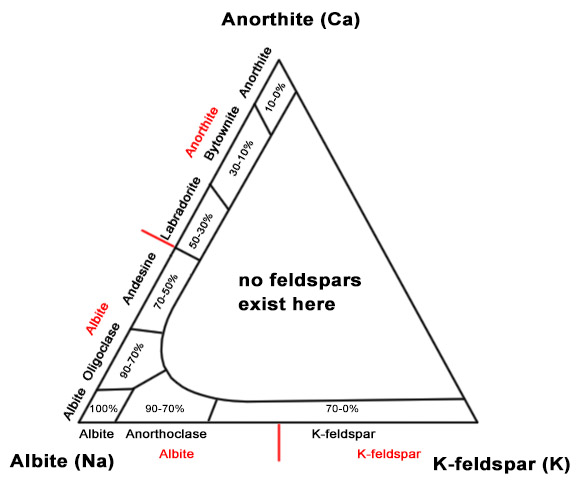
The plagioclase group consists of albite-anortite series of feldspars. With the 50% rule, all of the mineral members occuring between these two are not considered valid minerals by IMA. Basically, 50% rule says that Na dominant plagioclase is now albite and Ca dominant is anorthite. However, the old plagioclases names are still used in petrology for classification of rocks. The discarded plagioclase feldspars include oligoclase (10-30% of albite), andesine (30-50%), labradorite (50-70%) and bytownite (70-90%). Oligoclase and andesine are now albite, labradorite and bytownite are anorthite. Most of the old-classification plagioclases are very hard to distinguish from each other. The only exception is labradorite, which exhibits nice play of colors and is often used as decorative stone.

Albite (NaAlSi3O8) and anorthite (CaAl2Si2O8) form a complete solid solution, which means they can mix with each other in any ratio. However, anorthite miscibility with other alcalic feldspars is very limited. Pure anortite is quite rare and can be found only in some special ultramafic rocks. Ca-rich plagioclases are common in mafic volcanic rocks like basalts, diorite or gabbro. They are also present in common metamorphic rocks like gneisses or amphibolite. Albite is a bit more special, so it has its own description bellow.
Albite, Pericline and Cleavelandite
Triclinic albite (NaAlSi3O8) belongs to both plagioclases and alkalic feldspars. It is beacause its ability to produce solid solution with both sanidine (K-feldspar) and anorthite (plagioclase). Albite is typical in silica rich rocks, especially in pegmatites. High (disordered) albite is also quite typical for high temperature greisen deposits where it replaces some minerals or even creates albite zones. Typically, albite is white or very slightly yellow in color; occasionally it can be also clear.

Most pegmatites contain significant amount of albite, often in the form of nice crystals. Big masses of white or slightly bluish albite are called cleavelandite. Sometimes, late metasomatic albite creates zones in pegmatite which resemble coarse grained white sugar. This variety is called sugar albite or saccharoidal albite and it is often associated with gemmy beryls or lithium tourmalines.
Elongated prismatic crystals of albite from alpine clefts are called pericline. It is common on many Alpine sites, though not that common like adularia. Pericline is often associated with chlorite.
Sanidine
The disordered monoclinic K-feldspar (KAlSi3O8) is called sanidine. It often forms twinned crystals of white, light gray or yellowish color. Clear yellow sanidine is very rare.

Sanidine does not occur in pockets and nice crystals are very rare. It is usually present in volcanic rocks like trachyte, rhyolite or phonolite. Sanidine often forms bigger crystals in fine grained matrix of these rocks. It can form also during high temperature and low pressure contact metamorphism. Gemmy yellow sanidine (previously described as orthoclase) comes from Itrongay on Madagascar.
Orthoclase and Adularia
Orthoclase is structurally "semi-ordered" monoclinic feldspar which falls between sanidine and microcline. It is often gray, pale yellow, pink or white, rarely could be also clear and gemmy. It readily forms crystal twins like Carlsbad twins (intergrown), Baveno and Manebach twins.

Size 5,5 x 4 cm. Photo: Zbynek Burival
Adularia is usually referred as variety of low (ordered) orthoclase, but its structure is often mixed with microcline. Some adularia is orthoclase, some is already microcline. Typically, adularia forms white or nearly clear crystals of typical shape inside alpine clefts. The type locality of adularia is Adula Massif in Switzerland but it is common on hundreds of other Alpine localities.

The classic locality for great orthoclase crystals are pegmatites of Elba island and Baveno in Italy, pegmatites of Strzegom in Poland and many pegmatites in USA, Brazil, Russia, China, France, Myanmar and lot of other places. Great crystals of adularia are found on many classic localities in the Alps, but also in alpine clefts of Afghanistan or Pakistan. The highly valued Carlsbad twins of orthoclase crystals come from Karlovy Vary (Carlsbad) granites in Czech Republic.
Microcline and Amazonite
The most structurally ordered triclinic K-feldspar is called microcline. It is a common mineral in acid magmatic rocks like granites or pegmatites and it is also often found in some metamorphic rocks like orthogneiss. It is usually white, light gray, pale yellow or light pink colored. Microcline is very similar to orthoclase and these two are often confused. Rare Pb-rich variety with blue-green color is called amazonite. Amazonite is typical for NYF class pegmatites.

Size: 12 x 10 cm. Photo: Zbyněk Buřival
The great microcline crystals come from many pegmatites around the world, including those in Brazil, USA, China, Afghanistan or Mongolia. The worlds best amazonites come from the Teller Co. in Colorado. Other high quality amazonites are from Russia, Norway, Mongolia and Ethiopia.

Size 5 x 3 cm. Photo: Zbynek Burival
Moonstone
White or semi-clear feldspar with play of colors is often called moonstone. The definition of the moonstone is a bit uncertain - as well as its mineralogy. Some consider moonstone a variety of microcline or orthoclase. Some moonstones are also perthite of albite an orthoclase or variety of oligoclase. Moonstone is often used for jewelry in cabochons.

Great moonstones come from Sri Lanka, other places include Brazil, India, USA, Madagascar or Australia.
Celsian and Hyalophane - Rare Barium Feldspars
The monoclinic barium member of feldspars with the composition Ba2Al2Si2O8 is called celsian. Celsian can easily mix with orthoclase, sometimes the Ba-rich K-feldspar (with up to 15 % of celsian) is called hyalophane.
Ba-rich feldspars are very rare and most of them are connected with some Mn-skarns, marbles or baryte veins. Very unique hyalophane occurence is Busovaca in Croatia, where it formed great crystals in Ba-rich alpine clefts.






Comments