Ultraviolet Fluorescence in Minerals
Fluorescence occurs when a mineral absorbs ultraviolet radiation and immediately re-emits visible light. This phenomenon transforms ordinary-looking specimens into vivid displays of color under UV lamps. There is a decent amount of mineral collectors, who specialize on UV active minerals.
What is Ultraviolet Light?
Ultraviolet radiation occupies the electromagnetic spectrum between visible light and X-rays. The visible spectrum ends at approximately 380-400 nanometers in the violet range. UV begins immediately beyond this threshold and ends at 100 nm.
While the distinction appears sharp in definitions, human sensitivity to deep violet and near-UV varies individually—some people can perceive wavelengths slightly below 380 nm under certain conditions.
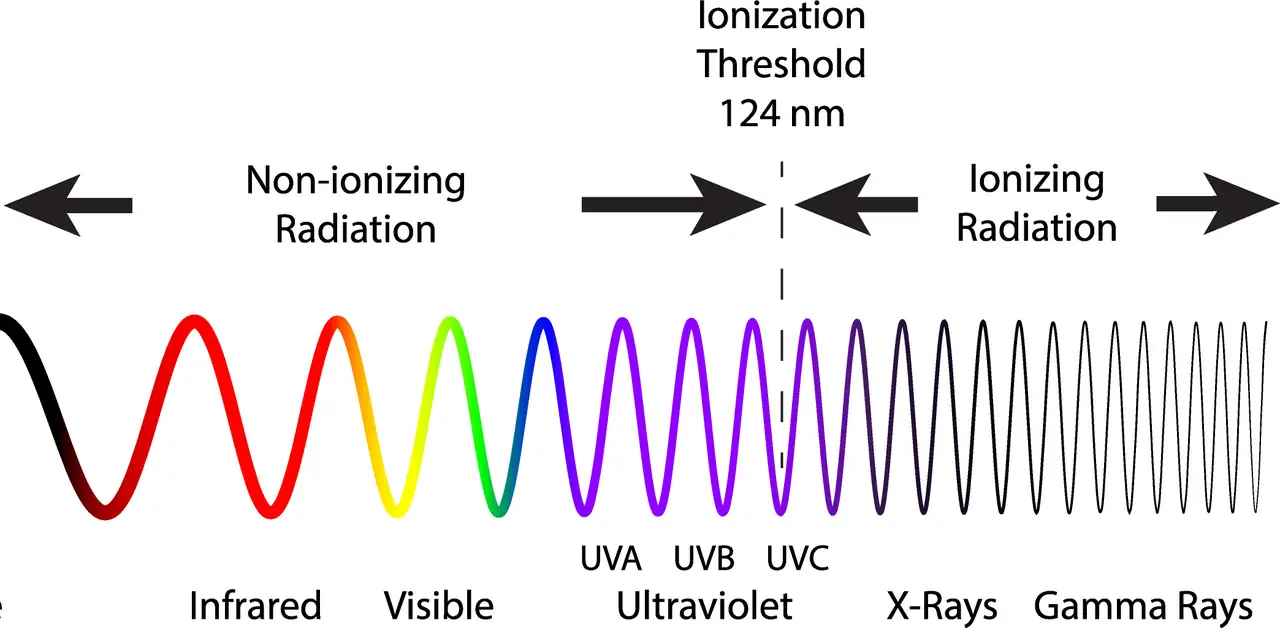
UV radiation subdivides into following categories based on wavelength and energy:
UV-A (315-400 nm) comprises the longest UV wavelengths, also called "longwave" or "blacklight". This is the least energetic UV and penetrates the atmosphere readily. UV-A passes through ordinary glass and constitutes most UV in sunlight reaching Earth's surface.
UV-B (280-315 nm) represents medium-wavelength UV, also called “midwave”. It is mostly absorbed by atmospheric ozone but partially reaching the surface. UV-B causes sunburn and has sufficient energy to damage biological molecules directly. UV-B is effectively blocked by glass.
UV-C (200-280 nm) includes the short and highly energetic UV wavelengths, also called “shortwave”. Atmospheric ozone absorbs UV-C completely, so natural UV-C never reaches Earth's surface. UV-C is germicidal and particularly hazardous to eyes and skin, requiring strict safety precautions during use.
VUV (100-200 nm) is the shortest UV, also called “vacuum UV”. It is absorbed by air, thus vacuum. This wavelength is used for lab experiments, but not for mineral collecting purposes.
Introduction to Luminescence
Generally, any type of glow caused by some kind of energy input without significant change of temperature is called luminescence. There is a wide spectrum of various luminescence types based on the energy source.
When energy input hits a luminescent mineral, it excites electrons in certain atoms to higher energy states. As these electrons return to their ground state, they release energy as photons of longer wavelength — typically in the visible spectrum.
This might sound complicated, but imagine core of the atom as the Earth and electrons as its satellites. The excitation means, that satellite (electron) moves to the upper orbital — this consumes energy. When the energy input is gone, the satellite (electron) drops back to the original lower orbital, and releases some energy back as a glow.
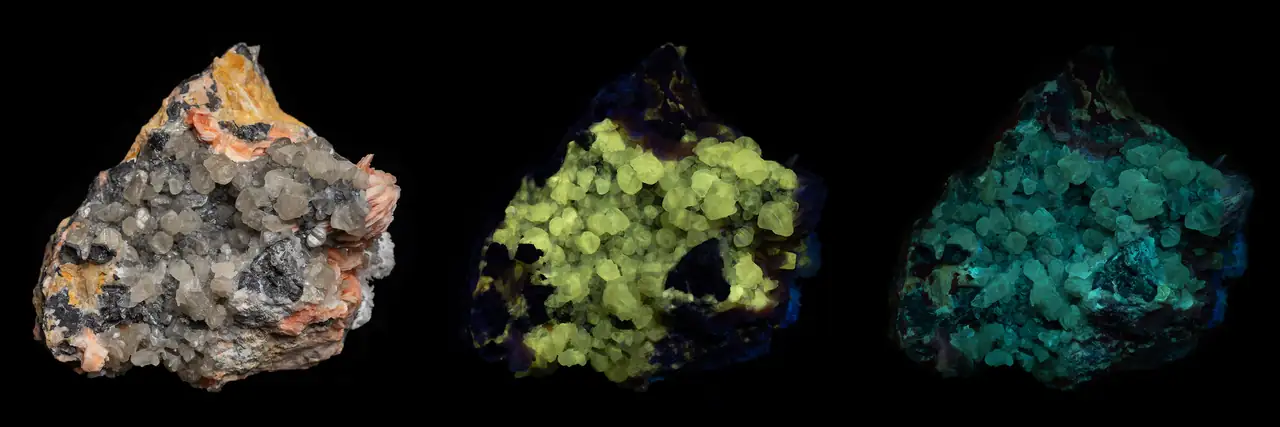
For the purpose of this article, we will consider only input of electromagnetic radiation — which is typically light. This is not limited to visible light, but includes way more interesting UV light as well.
There are 2 main interesting types of mineral luminescence, usually caused by UV light:
Fluorescence (or more generally photoluminescence) is usually used for instant glow of mineral specimens under UV light. Fluorescence ceases instantly when the UV light is removed.
Fluorescence is named after the mineral fluorite, in which the phenomenon was first systematically studied by G. G. Stokes around 1850.
Phosphorescence (or afterglow) is similar to fluorescence, but it is generally much weaker and persists for a while after the removal of UV light source. Afterglow might last from seconds to minutes or rarely even hours.
Ironically, phosphorescence is named after white phosphorus, which glows in the dark through a chemiluminescence from slow oxidation rather than true phosphorescence.
Fluorescence and Crystal Structure
Luminophores (or activators) are specific atoms, ions, or molecular centers within the crystal structure responsible for producing the observed glow.
These are typically the activator ions (like manganese, uranium, or rare earth elements) that absorb energy and re-emit it as visible light. However, the glow can be caused also by inclusions of fluorescent organic substances.
The produced glow color depends on the luminophore present and energy of incoming UV light.
The energy difference between UV wavelengths explains varying effects on minerals. Shortwave UV-C photons carry significantly more energy than longwave UV-A, sufficient to activate different fluorescent centers within the same mineral structure.

Not all specimens of a fluorescent mineral will glow. Luminophores are often present as impurities within a mineral structure or as inclusions.
Pure fluorite does not glow, but just small amount of REE’s turns it fluorescent. Presence of Mo changes the fluorescence color of scheelite, which varies from pale yellow to pale blue. Small amount or uranium renders feldspars or opals fluorescent.
What is Quenching?
The quenching agents (or quenchers) are substances or ions, which inhibit the UV fluorescence. Older mineralogical literature refers quenchers as killers, while general photochemistry literature might as well use terms like inhibitors or poisons.
The presence of quenchers — particularly iron in many cases — can completely suppress fluorescence even when activator ions are present. Quenchers absorb the energy provided by UV light and prevent radiation of a visible light (fluorescence).
UV Wavelengths and Mineral Response
Collectors typically use three UV wavelengths: longwave (365 nm) and shortwave (254 nm), much less common is midwave (310 nm).
Recent development made more exotic wavelengths available. This lead to interesting observations, where some minerals fluorescence under uncommon wavelengths. Or just a slight shift of common wavelength (like 10 nm) produces different fluorescence colors.
Many minerals fluoresce only under one wavelength (or rather some narrow range), while others respond to different wavelengths but with different colors or intensities.

Shortwave UV penetrates less deeply into minerals and is more energetic, sometimes exciting different activator centers than longwave. This is why a mineral might appear one color under longwave and another under shortwave, or fluoresce only under one wavelength.
Typical example is aragonite, which often produces different fluorescence colors under different wavelegths.
Overview of Common Fluorescent Minerals
There are many minerals displaying fluorescence. Following table is just a brief overview of most common fluorescent minerals:
| Mineral | Wave-length(s) | Color(s) | Cause |
|---|---|---|---|
| Aragonite | 365/254 nm | orange, pink, yellow | Mn, Pb, organic inclusions |
| Autunite | 365/254 nm | bright green | U |
| Calcite | 365/254 nm | red, orange, pink, purple, blue, yellow, white | Mn, Pb, organic inclusions |
| Corundum (var. Ruby) | 365/254 nm | red | Cr |
| Fluor-apatite | 365/254 nm | yellow, orange | REE |
| Fluorite | 365/254 nm | blue, purple, green, yellow, white, red, pink | Eu, Y, Ce |
| Gypsum | 365/254 nm | blue, green | organic inclusions |
| Opal (var. Hyalite) | 365/254 nm | bright green, white, pale blue | U, H2O |
| Quartz | 365/254 nm | bright green, white, pale blue | U, H2O |
| Scapolite | 365/254 nm | orange, red | S, Ce, Fe, Mn |
| Scheelite | 365/254 nm | yellow, blue, white | W, Mo |
| Sodalite | 365/254 nm | yellow, orange, red, white, pink | S |
| Sphalerite | 365/254 nm | blue, orange | Mn, Cu, Cd |
| Spodumene | 365/254 nm | blue, orange, purple | Mn, Eu, Dy, Ce |
| Willemite | 365/254 nm | bright green | ZnS |
Phosphorescence: The Afterglow
Phosphorescence differs from fluorescence in a crucial aspect: the light emission persists after the UV source is removed. This afterglow can last from fractions of a second to several hours.
The mechanism involves electrons becoming trapped in metastable energy states rather than immediately returning to ground state. These trap states are intermediate energy levels created by crystal defects or impurities.
Electrons gradually escape these traps through thermal energy, producing photons (phosphorescence) as they finally reach ground state. Temperature affects this release rate—warming a phosphorescent mineral can accelerate the glow, while cooling extends it.

Common phosphorescent minerals include certain willemites, some diamonds, and many synthetic zinc sulfides used in glow-in-the-dark products. Natural phosphorescence tends to be weaker and shorter-lived than synthetic materials, though exceptions exist. The duration and intensity of phosphorescence depend heavily on the concentration and distribution of trap sites within the crystal structure.
For collectors, phosphorescence adds another layer of identification. Testing requires darkness adaptation and patience, as subtle phosphorescence may take time to become apparent after UV exposure ceases.
UV Lamps and Flashlights
UV light sources vary considerably in quality, price and power. There are 2 main types:
Fluorescent Bulbs
In the past, the only reliable source were mercury vapor bulbs, typically producing 365 nm and 254 nm wavelegths (mercury emission lines). Much less common are 311 nm bulbs. All bulbs emit also significant amount of other UV wavelengths and visible light, thus the quality filter to cut off unwanted light is a must.

Big advantage of UV fluorescent bulbs is the high power and low price. Unfiltered bulbs can start at just $10, which makes them excellent choice for germicid lamps and other industrial use. Big setback is the size and power consumption.
Many UV displays use bulbs as the main light source. The big challenge of this approach is the necessity of active cooling and large expensive filters to cut off visible light.
UV LED
There was immense progress in the production of high quality UV LEDs in the last decade. UV LEDs quickly overtook the lead in portable UV lamps (flashlights), while bulky portable tube UV lights almost disappeared from the market. Some collectors already use LEDs even for their UV displays.
LED lights usually copy the well established wavelengths of bulbs, with the same longwave at 365 nm, but slightly shifted midwave at 310 nm and shortwave at 255 nm.
Modern development seriously improved the quality of the LED UV output, yet the power of LEDs is significantly smaller than bulbs. High quality LEDs are still quite expensive and require filters too.

The big advantage is the smaller size and lower power consumption. Smaller size of the light unit requires much smaller filters, which significantly reduces the price of the whole setup. While quality portable bulb lights often fall into $500-1000 range, cheap flashlights are available for $50 and really good ones for $300-400.
Photochemical Reactions in Minerals
Fluorescence can fade over time in some minerals, particularly those with organic activators. UV exposure may change structure of some bitumens, which renders them UV inactive.
Some minerals change their color after exposure to UV light, and slowly return to their original color. This effect is called tenebrescence or reversible photochromism, and is caused by a special structure and composition of some minerals. Common examples are sodalite (var. hackmanite), tugtupite, sometimes scapolite and spodumene.

There are also some minerals, which can change the color permanently due to the light and/or UV exposure. These changes are caused by photochemical reactions. Some of these changes are reversible (some spodumenes), and some are permanent (vivianite, silver minerals, realgar, pararealgar).
It is generally a good idea to keep light sensitive minerals in dark, not to mention exposing them to shortwave UV.
Safety and UV Light
Disclaimer: Hard UV light (particularly UV-B and UV-C) can be extremely dangerous and cause permanent health damage. Ensure you follow all safety precautions and instructions provided with your UV lamp or flashlight. The following text offers general recommendations and does not replace complete safety instructions, nor does it imply any liability, warranty, or responsibility for your actions!
UV-A tends to be underestimated, but 365 nm is already enough to irritate the naked skin or seriously irritate eyes. Unlike shorter wavelengths, it can partially pass even through the glass.
UV-B and UV-C are particularly dangerous for naked skin, as they easily cause sunburns and can potentially cause skin cancer. They are easily blocked by glass or plastic, but easily irritate or even damage your eyes!

Another unpleasant feature of shortwave UV is the production of ozone. This happens by reaction of UV light with atmospheric oxygen, producing typical odor. Various sources produce various amount of ozone, but even the no-ozone lamps produce some. Do not use shortwave lamps for a longer periods of time in closed rooms, ozone is toxic!
Ozone production from most UV flashlights is not a big issue and even UV bulbs are usually acceptable. But make sure to ventilate your room if you notice the ozone smell.
Using some cover for your skin (like textile gloves) and safety goggles is a must. We highly recommend some certified UV blocking goggles. Getting sunburns from shortwave UV is a matter of minutes!
References
- Bostwick, R. C. (2021): Thomas S. Warren (1903–2001): Godfather of the Fluorescent Mineral Hobby. Rocks & Minerals, 96(1), 76-82.
- Rakovan, J. (2021): Coming to terms with fluorescence: a short glossary of common terms in the context of mineral fluorescence. Rocks & Minerals, 96(1), 20-23.
- Robbins, M. A. (2013): The collector’s book of fluorescent minerals. Springer Science & Business Media.
- Root, J. (2021): Fluorescent Mineral Photography: A Shot In The Dark. Rocks & Minerals, 96(1), 69-75.
- Minershop/Mark Cole - Lot of articles, guides and reviews.
- NaturesRainbows.com - non-commercial website dedicated to fluorescent minerals.
- The Fluorescent Mineral Society - non-profit organization specialized on UV fluorescent minerals.





Comments